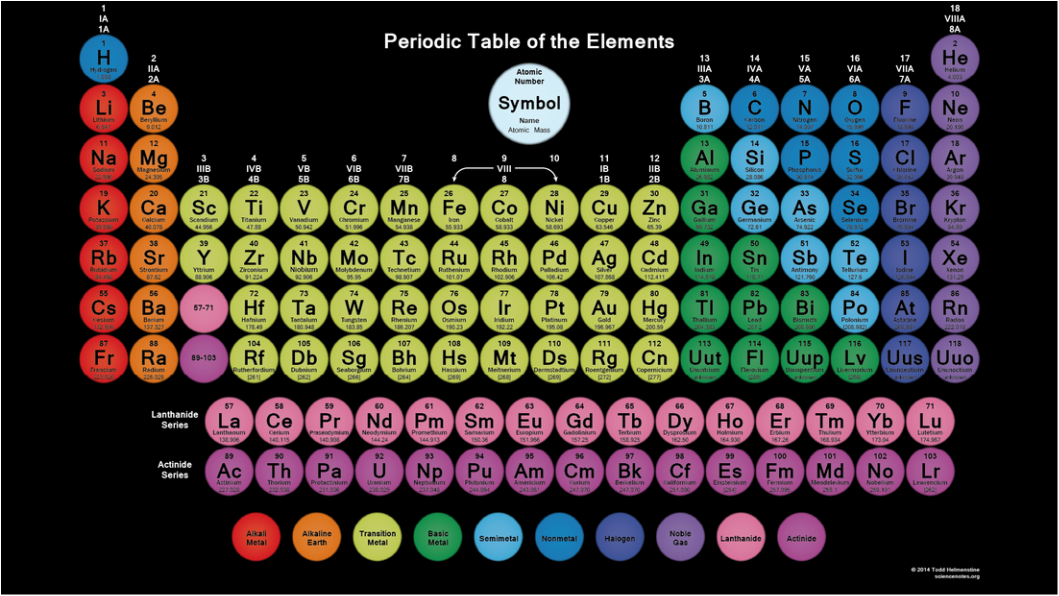
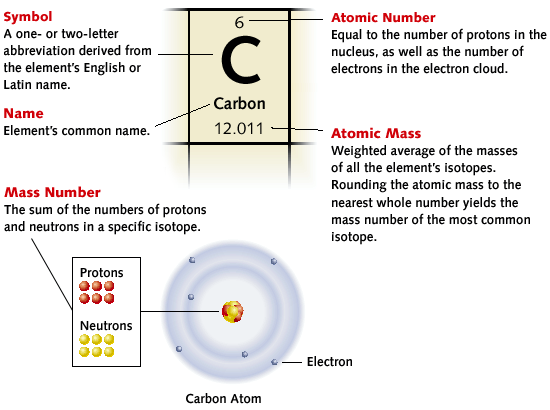
The Periodic Table of Elements is a table organized in a special way based on the atomic structure of an element.
How to Read a Box on the Periodic Table:
|
Groups
Groups are in columns on the periodic table. Each element in a group has the same number of valence electrons on the outer shell. The elements in the first column have 1 valence electron and likewise with the second column they have 2 valence electrons. It goes on like that as you count the columns except for the transition metals. |
Periods
The rows that read left to right are the periods. (Hence the name Periodic Table). The elements that are in the same period all have the same number of atomic orbitals. The first row has one orbital, the second row has 2 and it continues on that way down the periodic table. Currently, there is a maximum of 7 electron orbitals. |
The Different Families in the Periodic Table
|
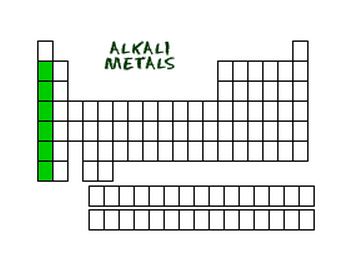
Alkali Metals
Alkali Metals are in the first group on the periodic table. Common properties are:
|
Hydrogen is not included in the alkali metals. They are very light weight which separates them from other metals. They are very soft and malleable. At times they can even be cut with a dull knife. You would not use these types of metals to make things such as buildings or hard money.
|
|
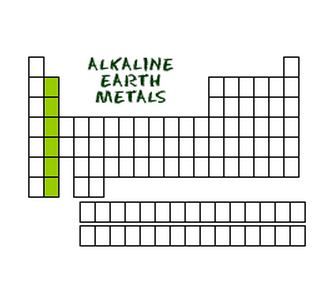
Alkaline Earth Metals
Alkaline Earth Metals are in the second group on the periodic table. Common properties are:
Transition Metals
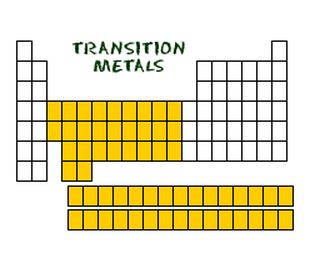
Transition Metals are from the third group to the eleventh group on the periodic table. Common properties are:
|
Transition metals are really special because they are able to put more than 8 electrons in the shell that is one from the outermost shell. The extra electrons are always added to the second outermost shell. Although most elements can only use their outermost shell in order to form bonds, the transition metals can use their two outermost shells. Most transition metals have two electron shells that aren't happy instead of just one.
Note: The maximum number of electrons in one shell is 32. |
|
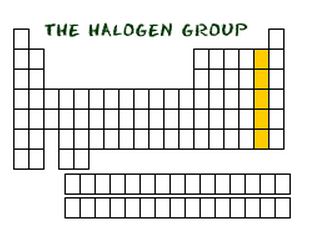
Halogen Gases
Halogens are from the seventeenth group on the periodic table. Common properties are:
|
|
Noble Gases
Noble gases are from the eighteenth group on the periodic table. Common properties are:
|
It is rare for noble gases to bond with other elements but it has been proven that they can bond with elements. Scientists have shown that this is possible by using these noble gases compounds to form explosives or just forming compounds in a lab. It is very rare for them to naturally form compounds though.
|
|
Note: The elements in groups thirteen through sixteen on the periodic table are a mixture of non-metals, metalloids, and metals.
|
Note: The twelfth group on the periodic table has a full d orbital shell and are all metals. The elements in this group tend to have low melting points.
|
Periodic Trends
|
Electronegativity
The electronegativity of an element increases as you go across a period from left to right. It decreases as you go down a group or family. As the number of electrons in an element's outer energy level nears a full octet then it gets much more difficult for an electron to be removed. The less shells of electrons the easier it is for a valence electron to be removed. Noble gases are not included. Note: Fluorine has highest electronegativity |
|
|
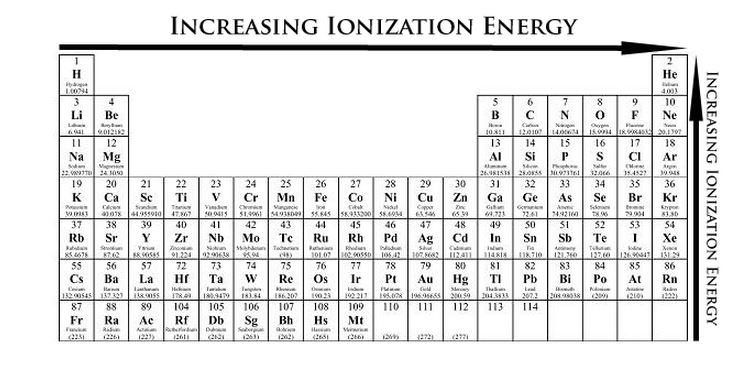
Ionization Energy
The ionization energy of an element is the energy required to remove an electron from a neutral atom in its gaseous phase. Ionization energy is the opposite of electronegativity. It increases as you go across a period from left to right and decreases as you go down a group or family. The higher the energy is, the less likely it is to become a cation. |
|
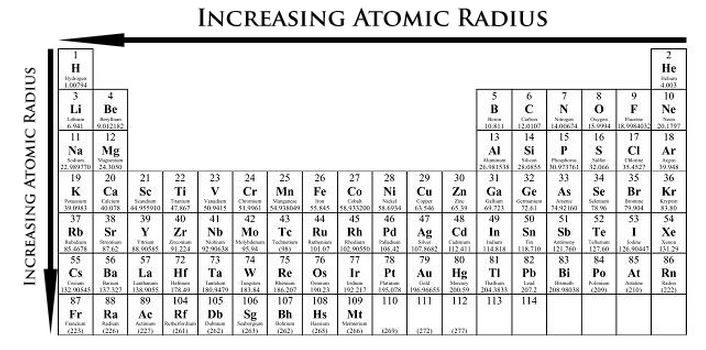
Atomic Radius
The atomic radius of an element is one-half the distance between the nuclei of two atoms. Atomic size gradually decreases from left to right across the periods. The reason for this is because when electrons are being added protons are also being added to the nucleus making it more positively charged. Since the increase of protons causes more of an impact than the increasing of electrons, the nuclear attraction is greater. Going down the group, the atomic radius increases also. Since the quantum number is increasing the valence electrons occupy higher levels. |