The Basic Structure of An Atom
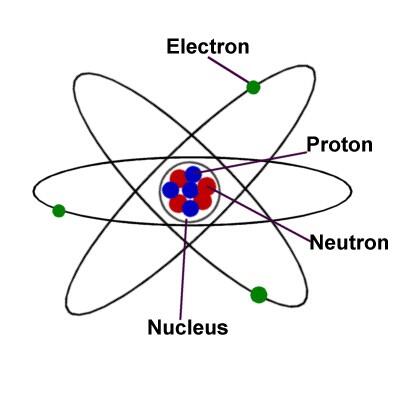
The basic structure of an atom contains the three subatomic particles:
|
Electrons
Electrons are the smallest part of the atom. They are found in shells or orbitals around the nucleus of an atom. They have a negative charge. The Nucleus is where the Protons and Neutrons reside.
|
Protons
Protons are subatomic particles that are found in the nucleus of an atom. They are large. They are positively charged. |
Neutrons
Neutrons are subatomic particles that are also found in the nucleus of an atom alongside the protons. They do not have a charge. |
Electron Orbitals and Configuration
Shells are the areas that surround the nucleus and there can be up to seven electron shells. The n is also used to say which shell number it is and at times, scientists will use the letters k through q in order to identify the other 7 shells.
|
Quantum Numbers
Quantum Numbers were developed in order to make it easier to understand the pathways of electrons in their designated orbital within an atom. |
Principle Quantum #: (n) Represents how far away electrons are from the nucleus the higher it is, the further away
Orbital #: (l) Determines shape of orbital Magnetic Quantum #: (m) Determines # of orbitals and their orientation in a subshell Spin #: (ms) Designates the direction of the electron's spin |
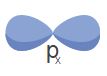
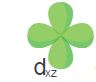
Suborbitals
Contributors to Atomic Theory
|
Democritus: Speculated that everything is made up of tiny particles called atoms.
John Dalton: Named the atom and said that they were like hard balls that cannot be split but can be joined together in order to make all materials. JJ Thompson: Discovered the electron. Said that they are divided equally throughout the atom. His model became known as the plum pudding model. Ernest Rutherford: Fired alpha particles at a gold plate and discovered the nucleus and protons. Niels Bohr: Believed that electrons orbited the nucleus in fixed layers which are called electron shells. (His model was based on only the Hydrogen atom) James Chadwick: Discovered neutrons. He found out that they weigh the same as protons but do not have a charge. Schrodinger
He disproved Bohr's model of the atom. His model is known as the quantum Mechanical Model. The rule states that no one can know the exact location of an electron but the location of the electron be predicted. Aufbau Principle His principle stated that the electrons take the most stable position and then go to the lowest energy level. Hund's Rule His rule was that the electrons will occupy different orbitals before sharing them (the electrons) |
|