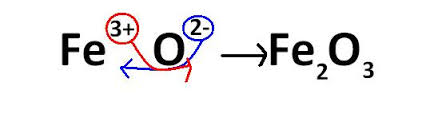
Ionic Naming
Compound to Name
*Write name of first element as it is originally. Change the ending of second element to "Ide"
Name to Compound
*Check charges on both elements and then switch them
Compound to Name
*Write name of first element as it is originally. Change the ending of second element to "Ide"
Name to Compound
*Check charges on both elements and then switch them
Acid and Base Naming
* If it is a Binary acid begin with "Hydro" and end with "ic" than add "acid"
(ex. HCl= Hydrochloric Acid)
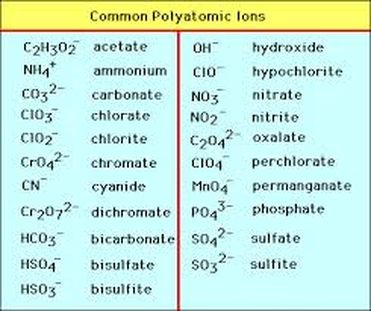
* If it is an acid with any polyatomic compound change the ending
If it ends in "ate" change to "ic" / If it ends in "ite" change to "ous"
Then add "Acid"
*Naming Bases is just like other compounds. Just don't forget the "hydroxide" or it is not a base anymore
* If it is a Binary acid begin with "Hydro" and end with "ic" than add "acid"
(ex. HCl= Hydrochloric Acid)
* If it is an acid with any polyatomic compound change the ending
If it ends in "ate" change to "ic" / If it ends in "ite" change to "ous"
Then add "Acid"
*Naming Bases is just like other compounds. Just don't forget the "hydroxide" or it is not a base anymore